Anti-Syntaxin 4 antibody
| Name | Anti-Syntaxin 4 antibody |
|---|---|
| Supplier | Abcam |
| Catalog | ab77037 |
| Prices | $385.00 |
| Sizes | 100 µg |
| Host | Mouse |
| Clonality | Monoclonal |
| Isotype | IgG1 |
| Applications | WB FC ICC/IF ICC/IF ELISA |
| Species Reactivities | Mouse, Human |
| Antigen | Recombinant protein fragment with GST tag, corresponding to amino acids 19 - 120 of Human Syntaxin 4 (NP_004595) |
| Description | Mouse Monoclonal |
| Gene | STX4 |
| Conjugate | Unconjugated |
| Supplier Page | Shop |
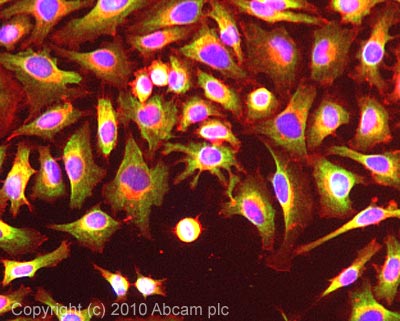
Product images
Product References
Insulin stimulates syntaxin4 SNARE complex assembly via a novel regulatory - Insulin stimulates syntaxin4 SNARE complex assembly via a novel regulatory
Kioumourtzoglou D, Gould GW, Bryant NJ. Mol Cell Biol. 2014 Apr;34(7):1271-9.
Abnormal myocardial insulin signalling in type 2 diabetes and left-ventricular - Abnormal myocardial insulin signalling in type 2 diabetes and left-ventricular
Cook SA, Varela-Carver A, Mongillo M, Kleinert C, Khan MT, Leccisotti L, Strickland N, Matsui T, Das S, Rosenzweig A, Punjabi P, Camici PG. Eur Heart J. 2010 Jan;31(1):100-11.
![Overlay histogram showing HL60 cells stained with ab77037 (red line). The cells were fixed with 80% methanol (5 min) and then permeabilized with 0.1% PBS-Tween for 20 min. The cells were then incubated in 1x PBS / 10% normal goat serum / 0.3M glycine to block non-specific protein-protein interactions followed by the antibody (ab77037, 1µg/1x106 cells) for 30 min at 22ºC. The secondary antibody used was DyLight® 488 goat anti-mouse IgG (H+L) (ab96879) at 1/500 dilution for 30 min at 22ºC. Isotype control antibody (black line) was mouse IgG1 [ICIGG1] (ab91353, 2µg/1x106 cells) used under the same conditions. Acquisition of >5,000 events was performed. This antibody gave a positive signal in HL60 cells fixed in 4% paraformaldehyde (10 min)/permeabilized with 0.1% PBS-Tween for 20 min used under the same conditions.](http://www.bioprodhub.com/system/product_images/ab_products/2/sub_5/7719_Syntaxin-4-Primary-antibodies-ab77037-2.jpg)